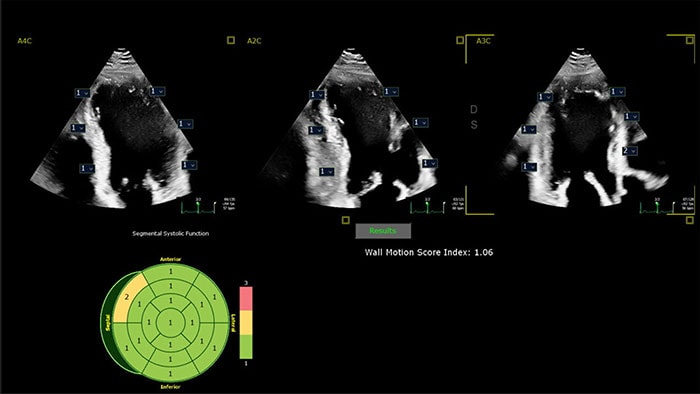
With the growing prevalence of cardiovascular disease, cardiologists are balancing the need to deliver high-quality care for a growing number of patients with complex conditions [1] while managing pressures to improve efficiency within their department or practice.
The ability to provide comprehensive, actionable insights into a cardiac patient’s heart health from their home via remote monitoring devices has proven to be a valuable tool for cardiologists as they seek ways to enhance cardiac care pathways. To provide an understanding of how ambulatory monitoring is changing cardiac patient care, Manish Wadhwa, MD, FHRS, Head of Medical Office, Ambulatory Monitoring and Diagnostics at Philips sat down with Dhanunjaya Lakkireddy, MD, FHRS, Executive Medical Director of the Kansas City Heart Rhythm Institute at HCA Midwest Health. The two practicing Cardiac Electrophysiologists discuss how the value of remote monitoring technology lies in its ability to provide clinically significant, actionable insight.

Dr. Lakkireddy

Dr. Wadhwa
Dr. Wadhwa: There have been several advancements in ambulatory monitoring technology over the past 20 years. How has the evolution of technology and new devices impacted the diagnostic and patient experience?
Dr. Lakkireddy: Three to four decades ago, when patients told their physicians they were having palpitations, there wasn’t a great way to capture cardiac arrhythmias in real-time, short of connecting the patient to an ECG machine in the hospital. As a result, it was harder for physicians to make accurate diagnoses. For example, many patients, particularly young women, were mislabeled as having “spells,” anxiety, or neurosis when they were actually experiencing Supraventricular Tachycardia (SVT). Over time, remote monitoring devices have evolved to provide physicians with more insight into how the heart is behaving, which is critical in informing timely interventions and confident diagnoses to support better patient outcomes.
The invention of the Holter monitor was a breakthrough in cardiology, giving physicians a more dynamic way to record arrhythmias. As healthcare’s digital transformation continues to take shape, access to patient data and dynamic monitoring has become increasingly more important – highlighting the limitations of Holter monitors’ microchip capacity and speed of data transmission. This paved the way for the next big leap in dynamic ambulatory monitoring through the live transmission of data via mobile cardiac telemetry, making it easier for us to seek immediate help for patients who were experiencing an adverse cardiac event.
In the past 15 years, we have seen a revolution in this space thanks to all the technological innovations in wireless and Bluetooth monitoring. This led to a rapid succession and evolution of these technologies alongside the miniaturization and increasing memory of the microchip, making physicians’ ability to record data significantly more efficient. The new monitors also greatly increased patient quality of life because their smaller size was much more portable and less invasive, and increased Bluetooth capabilities meant they could move freely without being connected to a wire.
Dr. Wadhwa: With the availability of connected devices that can monitor patients for extended wear times, how do you determine which monitoring device to use, and the duration based on the clinical scenario?
Dr. Lakkireddy: Today, there are three main types of monitors on the market – external monitoring devices, implantable long-term monitors and patient-driven consumer wearable monitors. Each type of monitoring device has its own advantages and disadvantages, which is why it is so important to pick the device that best suits the clinical scenario.
Decisions on which monitor to use are typically made on a case-by-case basis. However, there are several general parameters that shape these decisions. For example, for common disorders where a cardiologist may be more confident that a diagnosis can be made within four to six weeks, they are also likely more open to prescribing a mobile cardiac telemetry unit. With arrhythmias that are far apart and incidents that are unpredictable, an Implantable Loop Recorder (ILR) with backend remote monitoring support might be a better choice.
Choosing the best monitor for the patient really depends on what you’re looking for. For example, I used to use Holter monitors for Atrial Fibrillation (AFib) patients, but now I’ve changed my practice to use Philips Mobile Cardiac Telemetry – MCOT with remote monitoring services because it provides a more dynamic picture in terms of patient management. Remote monitoring with Philips MCOT gives physicians access to critical data about patients’ day-to-day life like atrial fibrillation burden, event duration, activity level, and symptoms. These data can help guide clinical decision-making on when to adjust patient medication, timing of ablation, or when to have them back for a check-up.

Dr. Wadhwa: Thinking about clinical scenarios and disease states where remote monitoring is critical, how does Philips MCOT support better clinical and financial outcomes while supporting the overall patient experience?
Dr. Lakkireddy: In many clinical trials, we have seen that the longer you monitor a patient, typically the better their outcome will be [2,3]. Four to six weeks of monitoring is more advantageous than the previously used 24–48-hour devices because the greater amount of data provides a more holistic picture and improved insight into how the patient’s heart is acting.
For a condition like cryptogenic stroke, the probability of finding an underlying atrial-arrhythmia as the perpetrator of a thromboembolic event is significantly higher when the patient is monitored for a longer period of time, which is where Philips MCOT units excel.
After a Transcatheter Aortic Valve Replacement (TAVR), complications typically present themselves in the first few days or weeks following the procedure. To provide proactive care and improve outcomes where patients are most comfortable, many hospitals have begun to leverage MCOT technology to monitor for significant arrhythmias beyond hospital walls. Prescribing MCOT to patients post-TAVR may provide an opportunity for better health outcomes or cost savings by monitoring for arrhythmic disturbances, and, if detected, enabling timely intervention via pacemaker and the avoidance of costly and dangerous emergency care.
Dr. Wadhwa: That’s very interesting. I agree that a longer duration of monitoring provides additive diagnostic yield. The 30 days of continuous monitoring on a non-invasive Philips MCOT unit as an alternative to using an ILR may represent a more cost-effective diagnostic strategy.
There has been a revolution around ILRs and direct-to-consumer remote cardiac monitors. What can the industry do to help care providers manage the data around these devices?
Dr. Lakkireddy: ILRs completely changed the way we treat arrhythmias. Continuous ILR monitoring gives physicians a much better picture of the patient’s lifestyle, which allows for real-time intervention in emergencies, more informed treatment decisions, and more precise care for the patient. One disadvantage of both ILRs and wearable consumer monitors is the data deluge that continuous monitoring creates. As many existing processing units are not positioned to aggregate extensive amounts of incoming data, providers must sift through this patient data manually.
To lessen the data burden presented by ILRs and direct-to-consumer monitors, new devices should focus on actionable insights. Care providers want to know about events that are important and require intervention, without being continuously alerted of incoming data from patients who are not experiencing any adverse symptoms. When it comes to monitoring, I always say, ‘The more you look the more you find, the more you find, the more you record, the more you record the more answers you have.’ If doctors don’t incorporate monitoring, there is less data to look at, but they may also be missing the answers needed to make a difference for the patient.
For cardiology departments and private practices, setting up new monitoring devices, it is common for devices to be used with their “out of the box” settings. However, to get the best results, I’ve found that it can be helpful to review available resources, focusing on the specific conditions they are treating, and indications for how these devices are being used together. This packaged view allows care providers to set and optimize guardrails for what clinically significant data should lead to notifications. In my practice, we have a “red, yellow, green” notification system to help us determine if a patient requires immediate intervention, whether they should be monitored more closely for a set amount of time, or if they do not require additional attention at that time. Having effective communication to understand the needs of the clinical care team who is treating patients every day is crucial to running an effective monitoring program.
Dr. Wadhwa: What other data points should we be working on as we advance connecting the care journey between hospital and home for cardiology patients to enable an accurate diagnosis and intervention at the right time and in the right setting?
Dr. Lakkireddy: The responsiveness of devices and the ability to support communication between patients and physicians in a timely fashion is critical. Much of the communication and responsiveness component is determined by staffing and the level of burnout on the monitoring team, which is why it is so crucial to meet as a department or organization and figure out how monitoring technology can best support the clinical environment.
Sometimes data points like heart rate variability and patient activity level are overlooked, but these are critical pieces to understanding the holistic picture of a patient’s health. The personal motto that I share with my department to motivate consistent check-ins with patients is, “when in doubt, monitor.”
[1] Journal of the American College of Cardiology, “Global Burden of Cardiovascular Diseases and Risk Factors, 1990–2019,” December 2022. [Online]. Available: https://www.jacc.org/doi/10.1016/j.jacc.2020.11.010
[2] New England Journal of Medicine, “Heart-Rhythm Monitoring for Evaluation of Cryptogenic Stroke,” 26 June 2014. [Online]. Available: https://www.nejm.org/doi/pdf/10.1056/NEJMe1405046
[3] ClinicalTrials.gov, “Long-term Cardiac Monitoring After Cryptogenic Stroke (CMACS) (CMACS),” 27 February 2014. [Online]. Available: https://clinicaltrials.gov/ct2/show/results/NCT00932425
Share on social media
Topics
Contact

Meredith Amoroso
Philips Global Press Office Tel: +1 724-584-8991
You are about to visit a Philips global content page
Continue