Philips teams up with leading innovators in interventional oncology
June 1, 2016
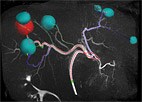
Amsterdam, the Netherlands – Royal Philips (NYSE: PHG, AEX: PHIA) today announced that as part of the existing Master Research Agreement with Yale School of Medicine, it has entered into a new research program led by Prof. Jeff Geschwind, chair of Radiology and Biomedical Imaging at Yale School of Medicine, to innovate in interventional oncology. Philips will collaborate with Prof. Geschwind and his team on a multi-year cancer research program to explore new concepts in image-guided therapies, as well as diagnostic imaging and informatics. The research program also involves further research groups at Yale School of Medicine. One of the corner stones of cancer care, medical imaging enables disease localization and quantification, therapy planning, and treatment guidance and assessment. Prof. Geschwind is a pioneer in interventional oncology, and following his recent appointment at the Yale School of Medicine, one of the top medical universities in the US, he has built an ambitious, comprehensive research program in cancer diagnostics and image-guided therapy. He has now teamed up with Philips as the program’s technology partner. Philips is a leading innovator in diagnostic imaging technologies, such as MRI, CT and Ultrasound, and associated health informatics tools that enable efficient data acquisition, integration and analysis. Moreover, the company has a broad portfolio of image-guided therapy solutions that enable clinicians to decide, guide and confirm the appropriate treatment. "Over the past few years there have been significant developments in image-guided therapy to locally treat tumors, with the result that interventional oncology procedure volumes have grown rapidly,” said Prof. Geschwind. “However, the biggest remaining challenge is that it is difficult to predict the effectiveness of the procedure. Together with Philips, we are embarking on a new multi-year research program with the aim to redefine and standardize this type of minimally-invasive treatment to achieve more predictable and better controlled procedure outcomes, and ultimately enhanced patient care.” “We are on a mission to enhance existing interventional oncology procedures and enable new ones that offer predictable and effective minimally invasive options for treating cancerous lesions,” said Bert van Meurs, General Manager Image Guided Therapy at Philips. “Our new collaboration with Prof. Geschwind and his team will bring together different scientific, engineering and medical specialties with the aim of developing, validating and translating breakthrough technologies for these targeted cancer therapies.” As part of the research agreement, a dedicated team of Philips clinical scientists will be working closely with Prof. Geschwind’s team at the Yale School of Medicine in a recently established state-of-the-art research facility optimized to translate laboratory results into enhanced patient care. Liver cancer, the second most lethal form of cancer [1], will be one of the focus areas in the collaboration. Only a minority of patients diagnosed with liver cancer are candidates for surgery, and intravenously administered chemotherapy has limited effect on the progression of the disease. An alternative technique known as transarterial chemoembolization (TACE) has shown better results than systemic chemotherapy and is now widely recognized as the best option for patients with inoperable advanced stage liver cancer [2]. TACE involves the local administration of chemotherapy agents and embolic beads that simultaneously block the arteries feeding the tumor and cause the tumor cells to die. The procedure involves the insertion of a catheter, which must be navigated to the tumor site with the aid of interventional X-ray guidance. However, the efficacy of TACE is often limited by the lack of predictive information prior to and during therapy – for example, information on the tumor biology and metabolism that is needed to determine the treatment strategy. Advanced diagnostic imaging and real-time image-guidance during the procedure, have the potential to advance TACE by consistently defining the best approach to the active tumor cells and confirming the distribution of therapeutic agents across the tumor. It is expected that ultimately this will not only improve the efficacy of the procedure, but will also encourage the more widespread adoption of new minimally invasive cancer treatment options. [2] Villanueva, A., Hernandez-Gea, V. and Llovet, J.M. (2013) Medical therapies for hepatocellular carcinoma: a critical view of the evidence. Nature Reviews Gastroenterology and Hepatology, 10(1), pp.34-42.
[1] http://www.who.int/mediacentre/factsheets/fs297/en/; Stewart BW, Wild CP. (2015) World cancer report 2014. International Agency for Research on Cancer, World Health Organization.
For further information, please contact:
Steve Klink Philips Group Communications Tel.: +31 6 10888824 E-mail: steve.klink@philips.com Philips Group Communications Tel.: +1 978-221-8919 E-mail: Kathy.oreilly@philips.com Twitter: @kathyoreilly
Kathy O’Reilly
About Royal Philips
Royal Philips (NYSE: PHG, AEX: PHIA) is a leading health technology company focused on improving people’s health and enabling better outcomes across the health continuum from healthy living and prevention, to diagnosis, treatment and home care. Philips leverages advanced technology and deep clinical and consumer insights to deliver integrated solutions. The company is a leader in diagnostic imaging, image-guided therapy, patient monitoring and health informatics, as well as in consumer health and home care. Headquartered in the Netherlands, Philips’ health technology portfolio generated 2015 sales of EUR 16.8 billion and employs approximately 69,000 employees with sales and services in more than 100 countries. News about Philips can be found at www.philips.com/newscenter.

