Jul 21, 2021
Philips receives FDA Breakthrough Device Designation for laser-assisted IVC filter removal device
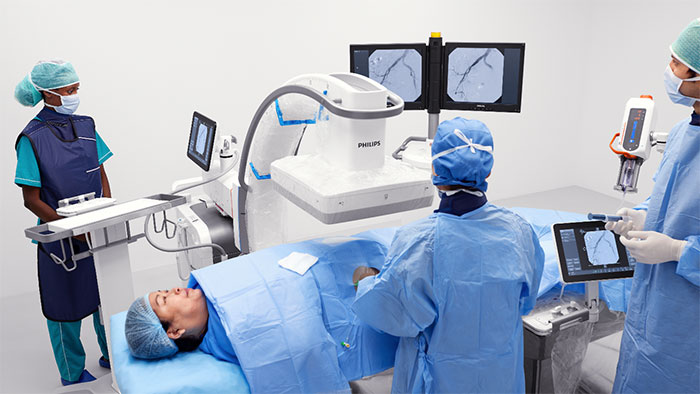
Amsterdam, the Netherlands – Royal Philips (NYSE: PHG, AEX: PHIA), a global leader in health technology, today announced the FDA has granted Breakthrough Device Designation (BDD) for a laser-assisted inferior vena cava (IVC) filter removal device. The proposed device is intended for ablating tissue to remove an IVC filter when previous methods of removal have failed. IVC filters are used to treat patients with venous thromboembolism, in which blood clots form in the deep veins of the leg, groin or arm and can travel through the circulatory system. They are placed in the inferior vena cava to capture blood clots from moving to the heart or lungs. Research has shown that IVC filters may have long-term complications [1]. The filters can fracture and travel through the bloodstream to other parts of the body. Other identified long-term risks associated with IVC filters include lower limb deep vein thrombosis and IVC occlusion. The FDA recommends that implanting physicians consider removing retrievable IVC filters as soon as they are no longer indicated [2]. The failure rate for IVC filter removal is high and limited options for removal exist if the filter has become difficult to remove. Advanced retrieval tools and techniques are required if the IVC filter becomes embedded in the vasculature [3]. Physicians have very few tools to remove the filter when this happens and there are no FDA-approved devices for this type of advanced removal.
Recent research supports laser-assisted removal
Two independent and prospective clinical studies demonstrated that laser-assisted retrieval was 96-99% effective with a major adverse event rate of 0.7-2% [3][4].
Breakthrough Device Designation reflects the potential impact that this technology could have on countless patients where this technology can be applied to safely retrieve filters by an expert physician, thereby reducing their risk of significant filter-related complications.
Kush R Desai
MD, FSIR, Associate Professor of Radiology, Surgery, and Medicine, and Director of Deep Venous Interventions at Northwestern University Feinberg School of Medicine, Chicago, Illinois, USA
“Laser-assisted filter retrieval may be a critical tool for the safe retrieval of filters with extended implantation times,” said Kush R Desai, MD, FSIR, Associate Professor of Radiology, Surgery, and Medicine, and Director of Deep Venous Interventions at Northwestern University Feinberg School of Medicine, Chicago, Illinois, USA. “Breakthrough Device Designation reflects the potential impact that this technology could have on countless patients where this technology can be applied to safely retrieve filters by an expert physician, thereby reducing their risk of significant filter-related complications.” “There is a clear need for an innovative device to help physicians more safely perform advanced IVC filter removal and I believe that the Philips excimer laser sheath may greatly enhance the options available to succeed in filter retrieval,” said Atul Gupta, MD, Interventional Radiologist and Chief Medical Officer, Image Guided Therapy, at Philips. “Breakthrough Device Designation is an important step for a medical innovation such as this one to help fill an unmet need in patient care.” FDA Breakthrough Device Designation is granted to novel medical devices that have the potential to provide more effective treatment or diagnosis of life-threatening or irreversibly debilitating diseases or conditions. The goal of the Breakthrough Devices Program is to provide patients and healthcare providers with timely access to breakthrough medical devices. Under the program, the FDA will provide Philips with priority review and interactive communication regarding device development throughout the submission process. Stay informed on developments – click here [1] Van Ha, T. G. Complications of inferior vena caval filters. Semin. Interv. Radiol. 23, 150–155 (2006).
[2] Health, C. for D. and R. Safety Communications - Removing Retrievable Inferior Vena Cava Filters: FDA Safety Communication. (2014). Available at: http://wayback.archive-it.org/7993/20170722215731/ https://www.fda.gov/MedicalDevices/Safety/AlertsandNotices/ucm396377.htm.
[3] Kuo, W. et al. Laser‐Assisted Removal of Embedded Vena Cava Filters: A First‐In‐Human Escalation Trial in 500 Patients Refractory to High‐Force Retrieval. Journal of the American Heart Association 9:24, 1-9 (2020). https://www.ahajournals.org/doi/pdf/10.1161/JAHA.119.017916
[4] Desai, K. et al. Excimer Laser Sheath-Assisted Retrieval of “Closed-Cell” Design Inferior Vena Cava Filters. J Am Heart Assoc; 9: e017240 (2020).
About Royal Philips
Royal Philips (NYSE: PHG, AEX: PHIA) is a leading health technology company focused on improving people's health and well-being, and enabling better outcomes across the health continuum – from healthy living and prevention, to diagnosis, treatment and home care. Philips leverages advanced technology and deep clinical and consumer insights to deliver integrated solutions. Headquartered in the Netherlands, the company is a leader in diagnostic imaging, image-guided therapy, patient monitoring and health informatics, as well as in consumer health and home care. Philips generated 2020 sales of EUR 17.3 billion and employs approximately 77,000 employees with sales and services in more than 100 countries. News about Philips can be found at www.philips.com/newscenter.
Topics
Contacts

Joost Maltha
Philips Global Press Office Tel: +31 6 10 55 8116
You are about to visit a Philips global content page
Continue
Fabienne van der Feer
Philips Image Guided Therapy Tel: + 31 622 698 001
You are about to visit a Philips global content page
ContinueMedia assets
Press releases
Get our press releases by e-mail
You are about to visit a Philips global content page
Continue