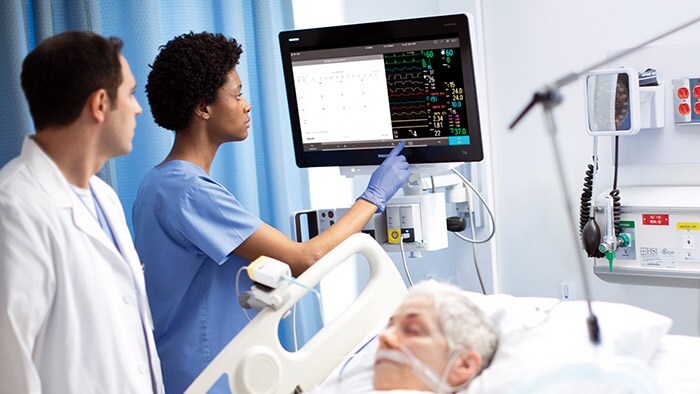
Philips welcomes the support of the U.S. government in increasing the production of hospital ventilators, which are critical for the treatment of COVID-19 patients. Philips’ employees are working around the clock in its US manufacturing sites to double production by May 2020 and achieve a four-fold increase by the third quarter of 2020. A vital element in this ramp up is ensuring a sufficient and constant supply of the 650+ different components that are needed for the assembly of Philips’ hospital ventilator. Philips is closely working with its suppliers in the US, Europe and Asia for an uninterrupted international flow of goods, which also requires the support of all the countries involved. The support of the U.S. government will strengthen Philips’ ventilator supplier base in the US, and help accelerate access to critical materials and components.
I am proud of the way American and Dutch companies are working together and stepping up their efforts in the fight against COVID-19.
Pete Hoekstra
United States Ambassador to the Netherlands
“I am proud of the way American and Dutch companies are working together and stepping up their efforts in the fight against COVID-19,” said Pete Hoekstra, the United States Ambassador to the Netherlands. “We are grateful to Philips for ramping up manufacturing on these urgently needed ventilator units. This is another example of the strength of the long-standing, unbroken ties between our two countries.” “We are actively cooperating with the U.S. government to expand our hospital ventilator production and reinforce our supply chain during these testing times,” said Frans van Houten, CEO of Royal Philips. “Our combined efforts will enable us to help healthcare providers in the US, the Netherlands and many other countries across the globe, with their challenging task to treat critical COVID-19 patients.” To facilitate the production expansion across its ventilator manufacturing sites in the US, Philips is investing several tens of millions in additional tools and molds, final assembly lines and test facilities. Moreover, the company is hiring extra manufacturing employees and changing to 24/7 shifts. Philips continues to actively engage with other governments around the world, the medical technology industry, the World Health Organization and other health authorities to safeguard the production expansion of materials, components and final products, as well as their shipment between countries.

Share on social media
Topics
Contact

Steve Klink
Philips Global Press Office Tel: +31 6 10888824
You are about to visit a Philips global content page
Continue