Philips wins the IEEE Spectrum Technology in the Service of Society Award
Philips Lumify, the world’s first app-based handheld ultrasound, empowers healthcare professionals to get diagnostic insight at the point-of-care, whether in the Emergency Department (E.D.) of a major hospital or at a clinic in a remote village. This groundbreaking and potentially life-saving innovation has been awarded the 2020 IEEE Spectrum Technology in the Service of Society Award – as the technology having the greatest potential to provide the most overall benefit to humankind. Judged by a panel of the publication’s editors, the award is presented for exceptional achievements in a wide variety of disciplines that have made a lasting impact on technology, society, and the engineering profession.
Transforming healthcare
Diagnostic images provided by an ultrasound transducer that is connected to a smartphone or tablet, and can be carried in a lab-coat pocket – Introduced in 2015, Lumify was the first app-based ultrasound system from Philips that opened up an entirely new category of diagnostic imaging devices. Since then, Lumify mobile ultrasound has transformed healthcare and supported life-saving procedures in the emergency department (E.D.), pre-hospital, and rural community settings. Bedside ultrasound imaging technology gives E.D. doctors access to real-time clinical information so that they can perform diagnosis of patients faster. In the ambulance, it supports physicians and emergency responders to diagnose and treat patients in the field, including to perform ECMO procedures, which could improve the chances of survival of cardiac patients by up to 30 percent. “Lumify has excellent image quality and is easy to use – it was instrumental in our first pre-hospital ECMO cannulation,” said Dr. Darren Braude, EMS Division Chief and a Professor of Emergency Medicine and Anesthesiology at UNM Health Sciences Center in the U.S.

Tele-ultrasound
Lumify with Reacts handheld tele-ultrasound solution lets clinicians around the globe collaborate remotely in real-time based on connectivity. While performing a scan on a patient, either device camera, voice, and live ultrasound image can be streamed to a healthcare professional remotely. The smartphone’s front-facing camera can be used to show the position of the transducer alongside the resulting ultrasound. The tele-ultrasound system can allow midwives in rural health facilities in Africa, to consult an expert ultrasound user regardless of the distance, who can provide perspective and guidance as if they were in the same room. During a 2019 project in collaboration with Imaging the World (ITW) in Uganda, mothers and newborns benefited from the scans. In 30% of the cases, the prenatal scans revealed high-risk conditions that initiated proper treatment and helped avert poor outcomes.
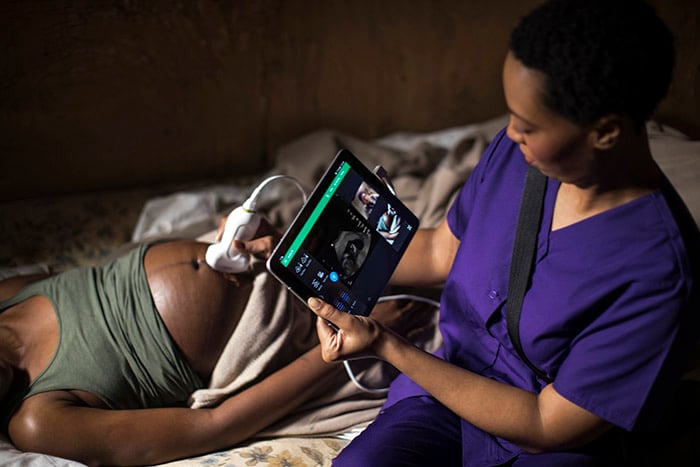
“The importance of Lumify’s high-quality ultrasound images must be emphasized. Without a clear picture, we can’t make correct diagnoses and then treat patients appropriately. This reliability is all the more critical in remote areas where geographic distance is a major factor in life vs. death for pregnant women,” said Dr. Kristen DeStigter, Professor of Radiology and Cofounder of Imaging the World. “Lumify not only delivers in this respect but meets the technical specifications necessary for remote practice in less-resourced areas. Lumify’s portability and ease of use promote improved access that positively impacts the wellbeing of women, children, their families, and their communities.” “It is an honor to be awarded by IEEE Spectrum for our Philips Lumify mobile ultrasound,” said Bich Le, Senior Vice President, and General Manager Ultrasound at Philips. “Lumify is a clear proof point of Philips’ dedication to expanding access to care by making ultrasound less expensive and easier to use.”
COVID-19 pandemic
Handheld and portable ultrasound solutions, in particular, have become valuable tools for clinicians treating COVID-19 patients due to their imaging capabilities, portability, and ease of disinfection. To support the global response to the COVID-19 pandemic, Philips has taken the necessary steps to obtain regulatory approvals in various markets impacted by this pandemic. More recently, Philips has received market clearance in several countries worldwide, including the U.S. Food and Drug Administration (FDA), CE Marking in the European Union, and approval from the Brazilian regulatory body to market Lumify and other ultrasound solutions for the management of COVID-19-related lung and cardiac complications. As a result of this regulatory clearance, Philips provides detailed, practical guidance to support clinicians using its systems and software for patients affected by COVID-19.
Commercially available in Japan
Philips received clearance from Japan’s healthcare authority to introduce Lumify with Reacts handheld tele-ultrasound solution in the world’s second-largest healthcare market after the U.S. Launched in Japan with a novel subscription model unique to the industry, Philips tele-ultrasound solution aims to make high-quality portable ultrasound available almost everywhere in the country.
About IEEE Spectrum
IEEE Spectrum is the flagship magazine and website of the IEEE, the world’s largest technical professional organization dedicated to advancing technology for the benefit of humanity. IEEE and its members inspire a global community through its highly cited publications, conferences, technology standards, and professional and educational activities.

Share on social media
Topics
Contact

Hans Driessen
Philips Global Press Office Tel: +31 6 10610417
You are about to visit a Philips global content page
Continue