Philips Titanion MR expands ultra-high-gradient MRI beyond anatomical and functional imaging toward whole-body quantitative biomarkers
May 11, 2026 | 3 minute read
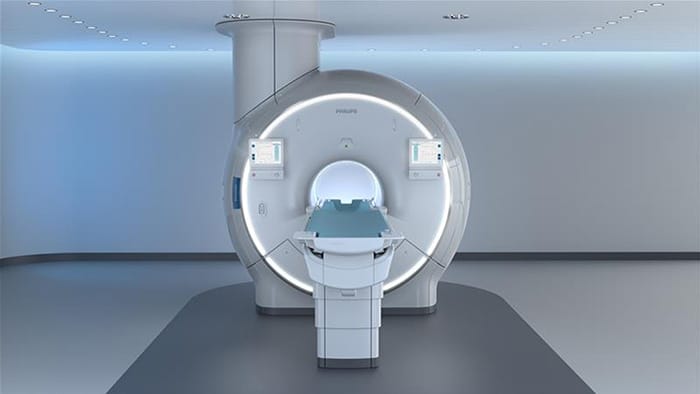
Amsterdam, the Netherlands – Royal Philips (NYSE: PHG, AEX: PHIA), a global leader in health technology, today announced Titanion MR, its ultra-high-gradient 3.0T MRI, at ISMRM 2026, the leading global forum for magnetic resonance research. Titanion is designed to advance MRI beyond anatomical and functional imaging by enabling clinicians and researchers to apply quantitative and biologically informative imaging approaches. By combining ultra-high gradient performance with AI-enabled workflows, it will support the development and application of imaging biomarkers that reflect tissue microstructure and underlying biological processes across the body.

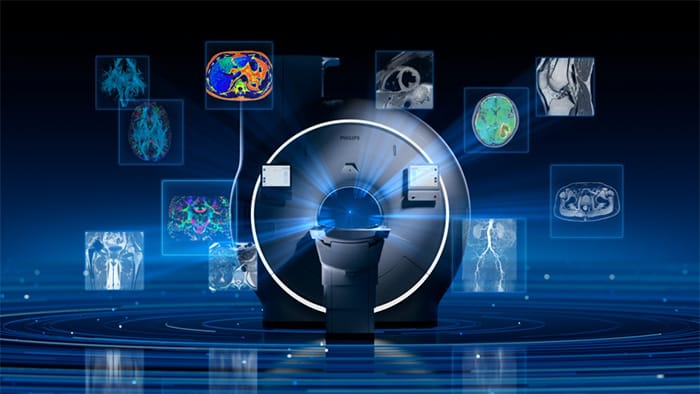
MRI continues to play an increasingly important role in diagnosis and disease monitoring. Philips Titanion MR is designed to advance this role by enabling imaging biomarkers that reflect tissue microstructure and function, supporting more precise and quantitative clinical insights across a range of applications, including neurology and oncology.
By combining ultra-high gradient performance with advanced AI-powered software, we are helping clinicians and researchers gain deeper insight into tissue microstructure and underlying biology, supporting the next phase of MRI toward more precise and quantitative results, enabling increasingly proactive and personalized clinical insights.
“Titanion MR represents a significant step forward for 3.0T MRI at Philips, expanding our ability to quantify imaging biomarkers across the body, especially in the brain, and bring advanced imaging capabilities into routine clinical practice,” said Ioannis Panagiotelis, Business Leader Magnetic Resonance at Philips. “By combining ultra-high gradient performance with advanced AI-powered software, we are helping clinicians and researchers gain deeper insight into tissue microstructure and underlying biology, supporting the next phase of MRI toward more precise and quantitative results, enabling increasingly proactive and personalized clinical insights.”
Built for breakthroughs in imaging biomarkers
Titanion is designed to bring ultra-high gradient performance to Philips’ 3.0T MRI portfolio, including:
Together, these capabilities will enable advanced quantitative and microstructural imaging, supporting the development and broader clinical application of imaging biomarkers and their translation into more precise, data-driven clinical care and research.
Intelligent performance for clinical practice
Titanion provides upgrades to Philips’ hardware innovation, combined with the company’s latest software advancements. With SmartSpeed, users can achieve up to 80% sharper images [3] and up to 3x faster scan times [4], supporting productivity and diagnostic confidence.
Titanion is designed with operational efficiency in mind, with power requirements of only 115 kVA [X], supporting installation in a wider range of clinical environments without extensive infrastructure upgrades, helping bring high-performance MRI capabilities to more sites.
Availability and configuration options for Titanion will depend on existing MR configurations and customer environments.
This webpage is intended for non-US audiences only. Titanion is a Work in Progress and not available in any jurisdiction. Its future availability cannot be guaranteed.
Sources [1] Titanion is considered Work in Progress and is not FDA cleared or CE marked and not available for sale.
[2] Pending verification
[3] Compared to conventional (SENSE / Compressed SENSE / SmartSpeed AI) imaging. Sharpness was evaluated with phantom scanning.
[4] Compared to Philips SENSE imaging.