Innovation Spotlight: Harnessing the speed of spectral CT
May 01, 2026 | 2 minute read
Spectral technology is expanding what is possible in computed tomography (CT), enabling clinicians to access richer diagnostic insights more quickly. Kevin M. Brown, Principal Scientist at Philips, shares his perspective on the evolution of spectral detector technology, the challenges behind bringing it to life, and how decades of innovation at Philips have shaped a new era in CT imaging.

Tell us a bit more about your background and what drew you to this specific role at Philips?
I like to invent things and solve technical problems, and my role allows me plenty of opportunities to exercise these skills. My undergraduate degree is in Electrical Engineering. I had the idea that I would like to get a PhD and teach, so I went to graduate school and chose to study Medical Imaging, because that seemed more interesting than building smaller cell phones (which were still quite bulky at that time). Creating a picture of the insides of a patient without having to open them up, is still kind of amazing. I did an internship with Picker (the company who owned the CT business at the time) during the summer, and they offered me a full-time position after graduation. A few years later Picker was purchased by Philips, and I’ve been with Philips ever since.
How does a CT scan work in general?
We take a series of 2D x-ray projections at various positions around the patient. The projections collapse 3D information into 2D, so that you cannot tell if one structure is in front of, or behind, another structure. A mathematical theorem called the Inverse Radon Transform allows us to use those series of 2D images to reconstruct a 3D volumetric image of the patient. This 3D image allows a doctor to truly see inside of the patient (at least for those structures and materials which have different stopping power for x-rays).

Can you outline the difference between a conventional CT scan vs. a spectral scan?
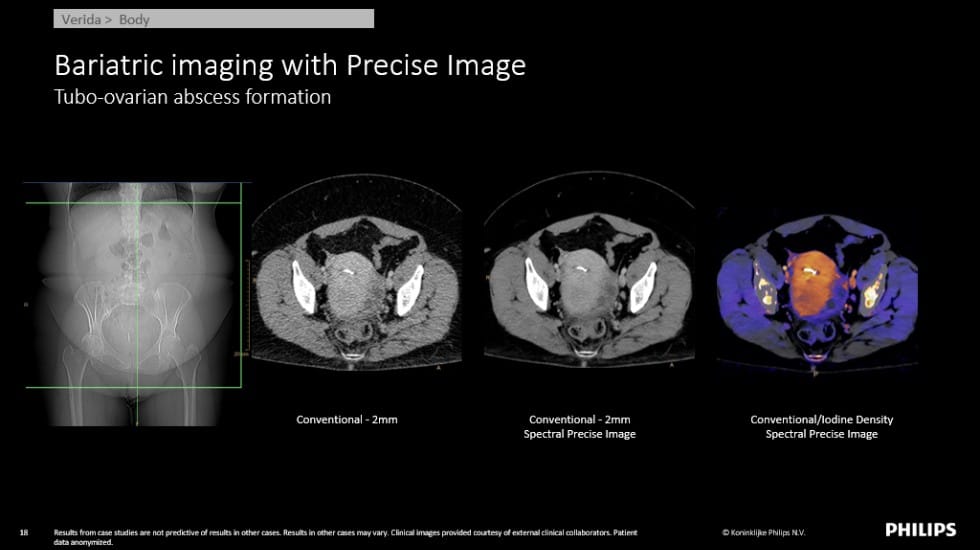
X-rays are emitted from an x-ray tube at a range of different energies. A conventional CT scan collects all of these energies together at the same time and uses them to make just one measurement. A spectral CT scan collects measurements of at least 2 different sets of x-ray energies (low-energy x-rays and high-energy x-rays, for example). Having these measurements at two different energies, allows us to estimate the attenuation properties of the different materials in the body over all x-ray energies, and this information allows us to distinguish between different materials, which may have been indistinguishable in a conventional CT scan. For example, calcium and iodine (which is used as a contrast agent injected into the patient’s bloodstream) may be difficult to separate on a conventional CT system, but with Spectral CT we can make specialized maps of which locations contain calcium and which locations contain iodine.
At the beginning with Spectral CT innovation, what drove you and made you so passionate about the project/technology?
There are different approaches to generating the dual energy measurements required for Spectral CT, each with its own advantages and trade-offs. With the dual-layer detector approach, one of the technical challenges early on was managing higher noise levels in the spectral images compared to conventional CT. This made it especially important to develop advanced reconstruction methods that can effectively handle this and still deliver clear, diagnostic-quality images. Solving that kind of complex technical challenge is what really motivated me.
What is the history behind Spectral CT at Philips? Can you explain how spectral detector technology was developed and the role of you/your team/the team (including timelines, testing, and validation if applicable)?
I didn’t personally have any role in detector technology development. The[KB3.1] principles behind Spectral CT have been known since 1976, shortly after the invention and demonstration of the earliest CT machines about 1973. But practical ways to acquire the different energies in a clinical setting (where the patient is often moving during the scan) took a long time to come into the market. Around 2005, a competitor developed a dual-source CT scanner (two x-ray tubes and two detectors) for the purpose of improving temporal resolution in cardiac CT scans. Having two x-ray tubes, they started to experiment with what else they could use them for, and they began to run the x-ray tubes at two different energies, and to advertise that these systems were also capable of Spectral CT imaging. But these dual-source systems had some compromises when operated in that mode, and customers had to plan a Spectral scan in advance while submitting to these compromises, and thus the adoption of Spectral CT in the routine clinical radiology practice was quite slow. Around the same time,[DE4.1] [KB5.1]Philips researchers at our detector center in Israel had been experimenting with dual-layer detectors as an alternative approach to Spectral CT, and our technical and marketing teams realized that we could build the first detector-based scanner with “always on” spectral capability, where users did not have to make compromises to enable Spectral CT. As a result, the plans for the first Philips Spectral CT system were made (the “IQon” scanner), which was released in 2015.
Were there specific innovations or design changes required along the way to enable or enhance spectral imaging compared to conventional CT systems?
The main hardware innovation was to build the dual-layer detectors which make it possible to make measurements at two different energy spectra at the same location and same point in time. The main software innovation was to come up with an elegant interface to allow users to interact with these now essentially 4D images (3 spatial dimensions, and one energy dimension) in an intuitive manner. And as mentioned above, we had to come up with new and improved reconstruction methods that could deal with the large increase in noise, and our computing team had to find ways to make sure all of these new reconstruction methods could be run fast enough to make the images at the same speed which customers were used to on conventional CT systems.
Can you describe the integration process between the detector, acquisition system, and software to enable spectral CT technology for the IQon, CT 7500 and then Verida—and what has been the biggest challenge?
The biggest challenge has been to help customers (radiologists) understand what they are seeing in a Spectral CT image, and how to use it to improve diagnosis and patient care. Since Spectral CT was a relatively new technology, many radiologists had no prior experience or training in what it was or how to use it. With much effort by our Clinical Science team over many years, in collaboration with key clinical partners, the benefits of Spectral CT are now widely recognized (with over 1000+ peer-reviewed papers demonstrating the importance and viability of detector-based spectral imaging in clinical practice to improve patient care)..[MD6.1] But as we continue to improve the quality of the Spectral Images, and remove more noise, we also continually encounter questions from customers about how to interpret these new things that they perhaps had difficulty seeing before. So, it’s an ongoing process and interaction between the developers of the technology and the end users.
How many people and what types of expertise are typically involved in bringing a fully functioning Spectral CT system to market?
Hundreds of people, with widely different skills including math, science, signal processing, engineering, mechanical and hardware, software, purchasing, finance, management, marketing, regulatory, and many others. It is truly a team effort, and everyone has their own important part to play.
How is AI influencing the development of Spectral CT, and what role do you see it playing in future innovation?
AI is a tool, and like any other tool, it is sometimes the right tool for the job, and sometimes not. As the capabilities of AI are continually expanding, I am sure that we will find new and innovative ways to incorporate AI into future CT products (from workflow to image quality). For example, we applied AI to reconstruction in new systems such as Verida Spectral CT to further improve the image quality, remove noise and improve low contrast detectability. Still, I’m also sure that we will continue to need “old-fashioned” tools like mathematics, problem solving, and human expertise.
If someone asked you, how would you describe in a simple way/analogy the difference between Spectral and conventional CT technology, how would you answer?
Conventional CT is like an old black-and-white television set. Spectral CT is like going from black-and-white TV to color television. With Philips Verida Spectral CT, the color TV is like going from an old analog over-the-antenna TV to a digital streaming television.